
StudySmarter - The all-in-one study app.
4.8 • +11k Ratings
More than 3 Million Downloads
Free
Americas
Europe
Remember learning the color wheel in art class? Combine blue and yellow, and you can get a shade of green. We say a “shade” of green because what you get depends on how much of each color you put together. Add a generous amount of red, and you might get a shade of brown. But add a tiny bit of red, and you might get a warmer shade of green.
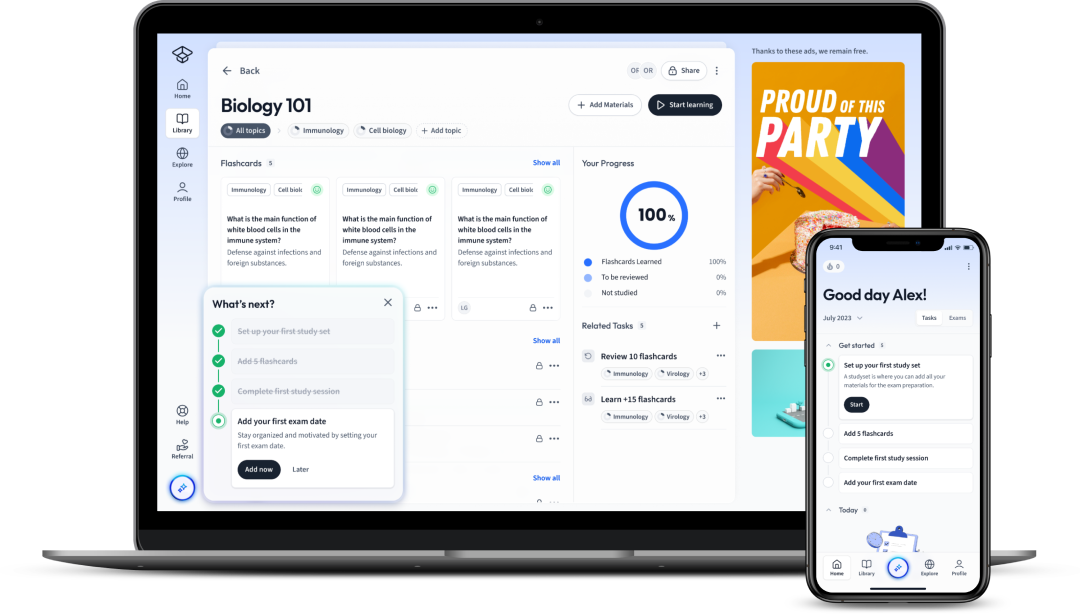
Explore our app and discover over 50 million learning materials for free.


























Lerne mit deinen Freunden und bleibe auf dem richtigen Kurs mit deinen persönlichen Lernstatistiken
Jetzt kostenlos anmeldenRemember learning the color wheel in art class? Combine blue and yellow, and you can get a shade of green. We say a “shade” of green because what you get depends on how much of each color you put together. Add a generous amount of red, and you might get a shade of brown. But add a tiny bit of red, and you might get a warmer shade of green.
The vast range of colors that we see around us can be reduced to three primary colors: blue, red, and yellow (note that this is not the case in physics!).
Now think of the different life forms that exist on Earth. From the tiniest bacteria to the massive blue whale, all organisms can be broken down into a few elements that are combined in varying proportions, structures, and through different chemical reactions. So, let's talk about the different elements of life!
All life forms are made up of matter, and all forms of matter are made up of varying combinations of elements. Elements are defined as fundamental units of matter that cannot be broken down or converted into other substances through ordinary chemical reactions. The smallest particle of an element that maintains its chemical properties is called an atom.
At present, there are a total of 118 elements: 92 of these elements occur in nature, whereas the rest are synthesized in laboratories and tend to be unstable (Fig. 1).
Matter refers to any substance that takes up space and has mass. It is made of a combination of elements.
Out of the 92 naturally occurring elements, only a handful make up all life on Earth.
Four elements are common to all living things: carbon (C), hydrogen (H), oxygen (O), and nitrogen (N). These four elements alone make up approximately 96% of all living matter. Sulfur (S), phosphorus (P), calcium (Ca), potassium (K), and a few other elements constitute the other 4% of an organism’s mass. Together, these elements are also sometimes referred to as bulk or major elements of life.
Elements found in living organisms are quite different from those of non-living things. For example, the atmosphere contains a lot of nitrogen and oxygen but very little carbon and hydrogen. On the other hand, the earth’s crust contains oxygen and hydrogen but contains only trace amounts of nitrogen and carbon.
In the following section, we will discuss how these elements combine in various ways to form compounds present in all living things. Specifically, we will discuss how these elements combine to form water and organic compounds.
Recall that all living things are composed of basic units called cells. A cell is primarily made up of water, which accounts for 70% of its mass. Keep in mind that intracellular processes also typically take place in an aqueous environment. This means that all life on Earth largely depends on the unique properties of water.
Water molecules are composed of two hydrogen atoms linked to an oxygen atom via a polar covalent bond. A covalent bond is formed when atoms share electrons in their outermost shell.
In a water molecule, the oxygen atom is highly electronegative, while the hydrogen atoms are less electronegative. This creates an uneven distribution of electrons, where there is a partially positive region on one side and a partially negative region on the other. This makes water a polar molecule.
Because it is a polar molecule, water molecules are able to form hydrogen bonds. Hydrogen bonding gives water molecules important life-sustaining properties including cohesion, moderation of temperature, and the ability to dissolve polar substances such as sodium chloride (also known as table salt).
Intracellular processes are processes that take place within the cell. These are said to take place within an aqueous environment because the cytoplasm (the fluid that fills the cell) is mainly composed of water.
In addition to water, cells are composed of carbon-based compounds that may contain up to 30 or so carbon atoms.
Carbon has an excellent ability to form large molecules: it has four electrons and four vacancies in its outermost shell, which means it can form up to four covalent bonds with other atoms.
Covalent bonds are chemical bonds that are formed between atoms that are sharing electrons.
Additionally, a carbon atom can attach to other carbon atoms through highly stable covalent carbon-to-carbon bonds that form chains and rings, allowing it to yield large and complex molecules. Such carbon-based compounds are called organic molecules.
Some of these organic molecules are monomers, which are simple subunits that bond together to form polymeric macromolecules. Other organic molecules are energy-rich substances that are broken down and converted into other smaller molecules in intracellular metabolic pathways.
You can consider a polymer to be a train made up of identical railroad cars, with each ‘car’ representing a monomer.
All organic molecules are made from and degrade into similar simple compounds. Both their synthesis and breakdown take place through sequences of chemical reactions that are restricted in scope and adhere to strict constraints. As a result, the compounds in a cell are similar in chemical composition, and the majority of them can be categorized as follows:
Carbohydrates are polymers composed of monosaccharides which are compounds made up of carbon, hydrogen, and oxygen with the general formula (CH2O)n, where n is typically a number from 3 to 8. An example of a monosaccharide is glucose (C6H12O6), an important source of energy for cells.
Lipids are polymers composed of fatty acids and glycerol. Fatty acids are made up of a hydrocarbon (C-H) chain and a carboxyl (-COOH) group. Glycerol is made up of carbon, hydrogen, and oxygen with the formula C3H8O3. An example of a lipid is the phospholipid, which is composed of a phosphate group, a glycerol, and two fatty acid chains (Fig. 2). Phospholipids make up the plasma membrane that encloses all living cells.
Proteins are polymers composed of amino acids. Amino acids are made up of a carboxylic acid group (-COOH), an amino group (-NH2), an organic R group or side chain, and a single carbon atom. Twenty types of amino acids are found in proteins, each with a different R group. These 20 amino acids are found in proteins, whether they are from bacteria, plants, or animals.
Nucleic acids are composed of nucleotides. Nucleotides consist of a nitrogenous base linked to a five-carbon sugar and a phosphate group. DNA and RNA, which contain the genetic information of all living organisms, are nucleic acids.
While there are many compounds found in cells that do not fall into these categories, these four families of organic molecules make up a significant portion of cell mass.
We have discussed how the four major elements (carbon, hydrogen, oxygen, and nitrogen), along with a handful of other elements (such as sulfur, calcium, and potassium) make up all living organisms.
However, there are some other concepts related to elements that could be worth noting. In this section, we will define essential and trace elements.
Out of the 92 naturally occurring elements, around 20-25% are considered essential elements that organisms need to survive and reproduce.
Organisms need similar essential elements, although in varying degrees. For example, humans require around 25 elements, while plants need only 17. Figure 1 below shows a list of essential elements in plants.
Note that these are categorized into macronutrients which are required in large amounts and micronutrients which are required in trace amounts (Fig. 3).
Macronutrients | Micronutrients |
| Required in large amounts | Required in trace amounts |
| carbon, phosphorus, nitrogen, hydrogen, potassium, magnesium, oxygen, calcium, sulfur | copper, iron, zinc, boron, manganese, molybdenum, nickel, chlorine |
Figure 3. This table shows the essential elements that plants require in order to grow and develop normally.
Without these essential elements, a plant may not be able to complete its life cycle: its seeds may not germinate, or it may be unable to form healthy roots, stems, leaves, or flowers. There are also possibilities that the plant may be unable to produce seeds at all. Worse, the plant itself could die.
While organisms require some elements in giant quantities (for instance, we have mentioned earlier that plants require macronutrients like carbon and phosphorus in huge amounts), they require other elements in minute quantities. The latter are called trace elements.
Some trace elements–like iron (Fe)–are required by all living organisms, while other trace elements are needed only by certain organisms.
For instance, vertebrates require iodine (I), an essential component of a hormone produced by the thyroid gland. In humans, 0.15 milligrams (mg) of iodine is required daily for the thyroid to function properly. A person deficient in iodine will suffer from a condition called goiter, wherein the thyroid gland grows to an abnormal size. This is why table salt is typically "iodized", meaning a small amount of iodine is added to it.
Zinc (Zn), copper (Cu), selenium (Se), chromium (Cr), cobalt (Co), iodine (I), manganese (Mn), and molybdenum (Mo) are all essential trace elements in the human body. Despite accounting for only 0.02 percent of total body weight, these components are crucial for certain biological processes, such as active sites of enzymes.
The elements that make up most life forms are carbon (C), hydrogen (H), oxygen (O), and nitrogen (N).
Five elements namely, carbon (C ), hydrogen (H), oxygen (O), nitrogen (N), and sulfur (S) make up most life forms.
Elements of life are the fundamental elements that make up living matter.
In addition to water, living matter are made up of carbon-based molecules. This is because carbon has an excellent ability to form large molecules: it has four electrons and four vacancies in its outermost shell, so it can form four covalent bonds with other atoms. Additionally, a carbon atom can attach to other carbon atoms through highly stable covalent Carbon-to-carbon bonds that form chains and rings which enable it to form large and complex molecules.
Flashcards in Elements of Life79
Start learningAcids give protons
True
Bases accept protons
True
Can a solution be an acid and a base?
Yes
What is an amphoteric substance?
A substance that can either be an acid or a base
What is a conjugate acid?
A base that gains a proton
What is a conjugate base?
An acid that loses a proton
Already have an account? Log in
Open in AppThe first learning app that truly has everything you need to ace your exams in one place
Sign up to highlight and take notes. It’s 100% free.
Save explanations to your personalised space and access them anytime, anywhere!
Sign up with Email Sign up with AppleBy signing up, you agree to the Terms and Conditions and the Privacy Policy of StudySmarter.
Already have an account? Log in