
StudySmarter - The all-in-one study app.
4.8 • +11k Ratings
More than 3 Million Downloads
Free
Americas
Europe
Explore the intricacies of antimicrobial resistance, a pressing issue in the field of microbiology, in this comprehensive guide. This exploration starts with a simplified explanation, leading onto its pivotal role in microbiology, and delving deeper into the biological mechanisms behind this phenomenon. Unveil the potential health and ecological risks linked to antimicrobial resistance and discover strategic solutions to mitigate its impact. Additionally, gain insights into the bond between antimicrobial resistance and communicable diseases. This thorough investigation into antimicrobial resistance is essential reading for every microbiology enthusiast.
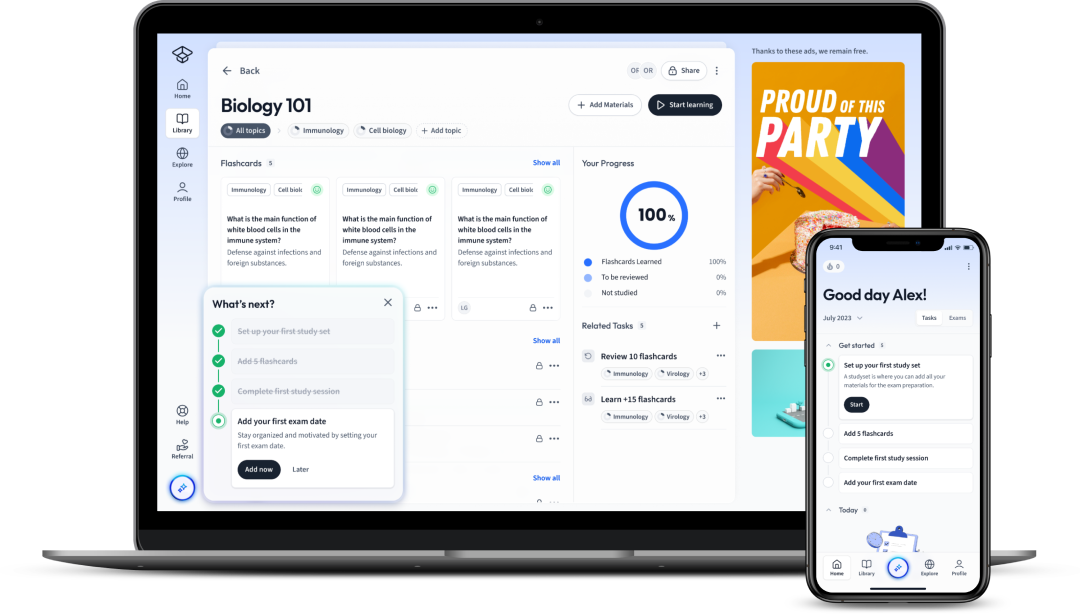
Explore our app and discover over 50 million learning materials for free.


























Lerne mit deinen Freunden und bleibe auf dem richtigen Kurs mit deinen persönlichen Lernstatistiken
Jetzt kostenlos anmeldenExplore the intricacies of antimicrobial resistance, a pressing issue in the field of microbiology, in this comprehensive guide. This exploration starts with a simplified explanation, leading onto its pivotal role in microbiology, and delving deeper into the biological mechanisms behind this phenomenon. Unveil the potential health and ecological risks linked to antimicrobial resistance and discover strategic solutions to mitigate its impact. Additionally, gain insights into the bond between antimicrobial resistance and communicable diseases. This thorough investigation into antimicrobial resistance is essential reading for every microbiology enthusiast.
A critical concept in studying microbiology, especially in the age of advanced medicine, is Antimicrobial Resistance. It refers to the phenomenon wherein microorganisms, bacteria and viruses primarily, become resistant to antimicrobial treatments, making it difficult to combat illness.
Antimicrobial resistance (AMR): The ability of a microbe to resist the effects of medication previously used to treat them.
To further simplify, imagine you have a bacterial infection and take a course of antibiotics. The medicine is designed to kill the infectious bacteria. However, not all bacteria get eradicated. Some may possess characteristics that render them immune to this particular antibiotic, allowing them to survive and reproduce. These are the antibiotic-resistant bacteria. This is the essence of antimicrobial resistance, which constitutes a significant health risk because diseases caused by such bacteria become harder to treat. They may need higher dosages or stronger antibiotics, both of which increase potential side effects.
Antimicrobial Resistance is not an event that just randomly occurs. Rather, it evolves over time through the processes of mutation and natural selection, which work at the genetic level in organisms to create attributes that provide survival advantages, such as antibiotic resistance.
For instance, in a population of bacteria causing pneumonia, some bacteria may have a random mutation that makes them immune to a particular antibiotic. When an infected patient is treated with that antibiotic, the susceptible bacteria die, but the resistant ones survive and multiply, creating a population of resistant bacteria.
Studying Antimicrobial Resistance is vital in the current global health context. The rise of superbugs, bacteria resistant to multiple antibiotics, threatens our ability to treat common infections, accelerating the urgency to understand and combat AMR.
Research into Antimicrobial Resistance allows us to develop new strategies and drugs to combat resistant bacteria. Furthermore, awareness helps in promoting responsible antibiotic usage, which reduces the selective pressure that accelerates resistance development.
| Significance | Relevance |
| Combat Superbugs | New Treatment Strategies |
| Responsible Drug Use | Prevents Resistance Development |
When considering microbial resistance, it's crucial to talk about the mechanisms through which this phenomenon gets its footing. The core biological processes driving antimicrobial resistance are not down to chance, but are an outcome of microbial evolution and survival.
There are several key biological principles at play within antimicrobial resistance mechanisms. These include mutation, natural selection, horizontal gene transfer, and mechanisms of drug inactivation.
Mutation: A change in the DNA sequence of a microbe’s genome, often resulting in an altered protein product. This might confer resistance to an antimicrobial drug.
Natural Selection: A process that leads to the survival and reproduction of organisms best suited for their environment, such as resistant microbes surviving in an antibiotic-rich environment.
Horizontal Gene Transfer: A process through which bacteria transfer DNA containing resistance genes to other bacteria, boosting resistance spread.
β-lactam antibiotics include penicillin derivatives, cephamycins, and carbapenems. The prescribed antibiotic attacks the bacteria's cell wall, leading to the microbe's death. Yet, some bacteria produce Beta-Lactamase enzymes which inactivate the antibiotic, making the bacteria resistant to treatment.
Just as in bacteria, viruses can also exhibit resistance to antiviral drugs. One of the most well-known examples of this is the Human Immunodeficiency Virus (HIV).
Antiviral Resistance: When viruses undergo genetic change (mutation) that decreases the effectiveness of drugs previously used to treat them.
The basic principles of mutation and natural selection apply here as well. But unlike bacteria, viruses do not reproduce by cell division; instead, they replicate inside the cells of their host using the hosts machinery.
The main driver of antiviral resistance is the high rate of mutation during viral genome replication. Coupled with the short generation time, this leads to the rapid evolution of drug-resistant viruses.
Another significant fact to note is that viruses can also carry out reassortment (in case of segmented RNA genomes) or recombination, where they exchange genetic material when two different strains of a virus infect a single cell. This can lead to the creation of a new subtype with a different resistance profile, further complicating treatment strategies.
Various strategies, from high barrier to resistance drugs, combination therapies to vaccination, are employed to manage and mitigate antiviral resistance.
| Antiviral Strategy | Rationale |
| High Barrier to Resistance Drugs | Some antiviral drugs require several significant mutations for resistance to develop, slowing the development of resistance. |
| Combination Therapies | Using multiple drugs targeting different viral functions can prevent resistance by requiring the virus to develop multiple resistances simultaneously to survive. |
| Vaccination | Vaccines stimulate immunity without the risk of resistance development. They reduce disease prevalence and, consequently, the chance of resistance development. |
Antimicrobial resistance is more than just a biological concept. It carries real-world consequences that affect both human health and the safety and stability of our ecosystems. Delving deeper into these issues helps highlight the gravity of antimicrobial resistance and fuels the need for attentiveness and action.
On the health front, the implications of antimicrobial resistance are staggering. Diseases once easily treatable can once again become severe or deadly, thanks to resistance. With antimicrobial medications becoming less effective over time, the risk of complications and prolonged recovery times increases. Therefore, even routine surgeries and minor infections could turn into major medical challenges.
Prophylactic: Treatments given or actions taken to prevent diseases.
In terms of ecological risks, antimicrobial resistance adds another layer of complexity to the diversity and sustainability of our ecosystems. One key issue arises from our water systems where an extensive range of antibiotics are introduced. Here, they exert a selection pressure on bacteria, not unlike the ones observed in a healthcare setting. As a result, water and soil ecosystems can harbour resistant bacteria, disrupting balance and posing further risk to health.
To truly grasp the scale of the health and ecological risks of antimicrobial resistance, it helps to understand the global context. Antibiotic resistance is not a contained threat; it knows no borders and can spread across countries and continents.
Globalization: The process by which businesses or other organizations develop international influence or start operating on an international scale.
With globalized travel and food supply chains, resistant bacteria can rapidly disseminate globally. If a person carrying drug-resistant bacteria travels to another part of the world, the bacteria travel with them. If they fall sick and the bacteria are passed onto another person, local antibiotics might not affect these foreign bacteria.
The risk is particularly high in areas where antibiotic use is not regulated, and medications are often available without prescription. In such countries, misuse and overuse of antibiotics are common, creating an ideal environment for the emergence and spread of resistant bacteria.
In this context, the World Health Organization (WHO) and other international bodies have recognized Antimicrobial Resistance as a global public health threat. This recognition has prompted coordinated efforts. Yet, despite efforts like the Global Antimicrobial Resistance Surveillance System (GLASS), difficulties in gathering and sharing information across borders remain a key challenge in this fight.
Global climate change could also exacerbate the ecological risks associated with antimicrobial resistance. Changes in temperature, precipitation patterns, and humidity can affect resistance gene flows in microbial communities. Climate change may also drive increased use of antibiotics in agriculture due to more frequent and severe outbreaks of livestock disease, contributing to the burden of resistance.
| Global Risk Factors | Impact |
| Unregulated Drug Use | Environments with extensive antimicrobial misuse and overuse foster resistance. |
| International Travel | Movement of people across borders enables dissemination of resistant strains. |
| Food Supply Chains | Trade in goods, especially livestock and farming produce, can spread resistant organisms over large distances. |
| Climate Change | Changes in global climate patterns can alter microbial communities and enhance resistance gene flows. |
Antimicrobial resistance is a significant public health concern that necessitates strategic and sustainable prevention methods. Exploring these diverse tactics helps to understand the different facets of problem-solving involved in addressing this worldwide health threat.
To battle the tide of antimicrobial resistance, a multifaceted approach is essential. This involves combining public health measures, healthcare policies, and individual behavioural changes. Each element targets a distinct facet of the issue whilst collectively they aim to reduce the incidence of resistance.
A crucial aspect of public health measures is effective antibiotic stewardship. This term refers to coordinated efforts to promote the appropriate use of antibiotics: ensuring the right drug, at the right dose, at the right time, for the right duration. This minimises unnecessary use, thereby reducing the selective pressure that fosters the development of resistance.
Antibiotic Stewardship: A coordinated program that promotes the appropriate use of antibiotics to improve patient outcomes, reduce microbial resistance, and decrease the spread of infections caused by multidrug-resistant organisms.
Surveillance systems to monitor antibiotic resistance are an integral part of public health measures. These collect data on the prevalence of resistant bacteria and changes in their distribution. This information guides the rational use of antimicrobial drugs, informs policy decisions, and helps assess the effectiveness of preventative measures.
Public health measures also target the prevention of transmission of resistant organisms. This can involve the use of infection control measures such as hand hygiene, use of personal protective equipment, and sterilisation of medical equipment. Vaccination programs also contribute by reducing the spread of infections, thereby lessening the need for antibiotic use.
Improved diagnostic techniques can help prevent antimicrobial resistance. By ensuring that antibiotics are only used when needed, and that the right antibiotic is chosen for a particular infection, excess use can be minimised. Rapid diagnostics for bacterial identification and susceptibility testing are invaluable tools in this regard.
Education is a powerful weapon against antimicrobial resistance. By increasing awareness of the risks associated with antibiotic misuse and understanding of when these drugs are necessary, healthcare professionals and the public can be empowered to make responsible decisions about antibiotic use.
For healthcare professionals, this might involve education on the principles of antibiotic stewardship, while for the public, the focus might be on understanding why antibiotics are not applicable for viral infections like the common cold.
Last, but certainly not least, are preventive strategies targeting the use of antibiotics in agriculture. Antibiotics are often used in high quantities in livestock farming, both as a disease prevention measure and a growth promoter. This extensive usage contributes significantly to the development and spread of antibiotic resistance.
Interventions can include far stricter regulation of antibiotic use in farming, with bans on certain particularly high-risk uses (such as the use of antibiotics as growth promoters). Increased surveillance of antibiotic resistance in agricultural settings, improved infection control, and measures to reduce the need for antibiotics (such as improved animal vaccination) can also form part of a comprehensive prevention strategy.
Together, these public health measures and preventive strategies offer a robust line of defence against the onset and spread of antimicrobial resistance.
Antimicrobial resistance and communicable diseases are deeply intertwined. In simple terms, when microbes, such as bacteria, become resistant to antibiotics, treating infections becomes more challenging. This escalates treatment costs, prolongs illnesses, and increases mortalities - thereby magnifying the burden of communicable diseases on public health systems around the world.
The development and spread of antimicrobial resistance bring about a significant change in how communicable diseases are managed. The loss of effective antibiotics allows common infections to persist and spread more freely within a community, potentially leading to outbreaks of diseases that were once easily controlled. Due to the mobility of today's global population, these risks aren't localized to a single community or country - they're a global threat.
Several communicable diseases, including tuberculosis, gonorrhoea, and pneumonia, are becoming harder to treat because of increased antimicrobial resistance. In most cases, the source of the problem lies in the misuse of antimicrobials.
Communicable Diseases: These are illnesses caused by a specific infectious agent or its toxic products. They can be transmitted directly or indirectly from an infected person, an animal or the environment.
The ecological factor also plays a role here. Environs such as hospitals can often become 'hotspots' for antibiotic-resistant bacteria, mainly due to the high use of antibiotics. These environments can, therefore, act as reservoirs for these resistant organisms, posing a risk to both community and hospital populations.
To combat the growing threat of antimicrobial resistance, holistic and innovative approaches are necessary. These approaches should encompass medical, behavioural, and institutional strategies.
For instance, the scientific community continues to explore alternative antimicrobial strategies. These include the development of new antibiotics, the use of antibodies to target bacteria, and the manipulation of microbiome communities.
Additionally, behavioural changes remain central to antimicrobial stewardship. This includes patient education on the proper use and disposal of antibiotics, as well as healthcare provider training on the principles of judicious antimicrobial use.
Finally, solutions should encompass institutional changes. Hospitals, for instance, need stringent infection control measures to prevent the spread of resistant bacteria within their wards. Likewise, global healthcare policies need to limit the use of important human antibiotics in livestock production—a currently rampant practice contributing to resistance.
In conclusion, tackling the intersection of antimicrobial resistance and communicable diseases is no small feat. It requires both the continuous development of innovative solutions and sustained commitment from individuals, institutions, and governments. But, it is a battle that holds the key to maintaining the efficacy of our antimicrobial drugs and preserving global health.
The first learning app that truly has everything you need to ace your exams in one place
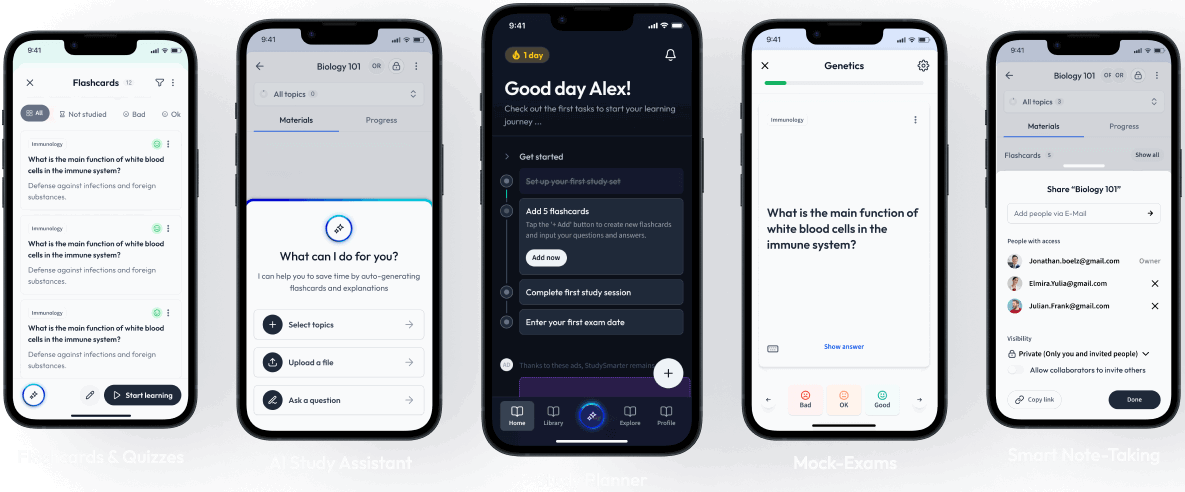
Sign up to highlight and take notes. It’s 100% free.
Save explanations to your personalised space and access them anytime, anywhere!
Sign up with Email Sign up with AppleBy signing up, you agree to the Terms and Conditions and the Privacy Policy of StudySmarter.
Already have an account? Log in