
StudySmarter - The all-in-one study app.
4.8 • +11k Ratings
More than 3 Million Downloads
Free
Americas
Europe
There is a continuous cycling of materials in the ecosystem. In the environment, waste and dead materials are continually recycled to support various life forms. This is not just limited to organic materials. Both organic and inorganic minerals are continuously exchanged and moved in the Ecosystems to support the production of matter. This article will discuss the three most essential mineral cycles and their importance in ecosystems.
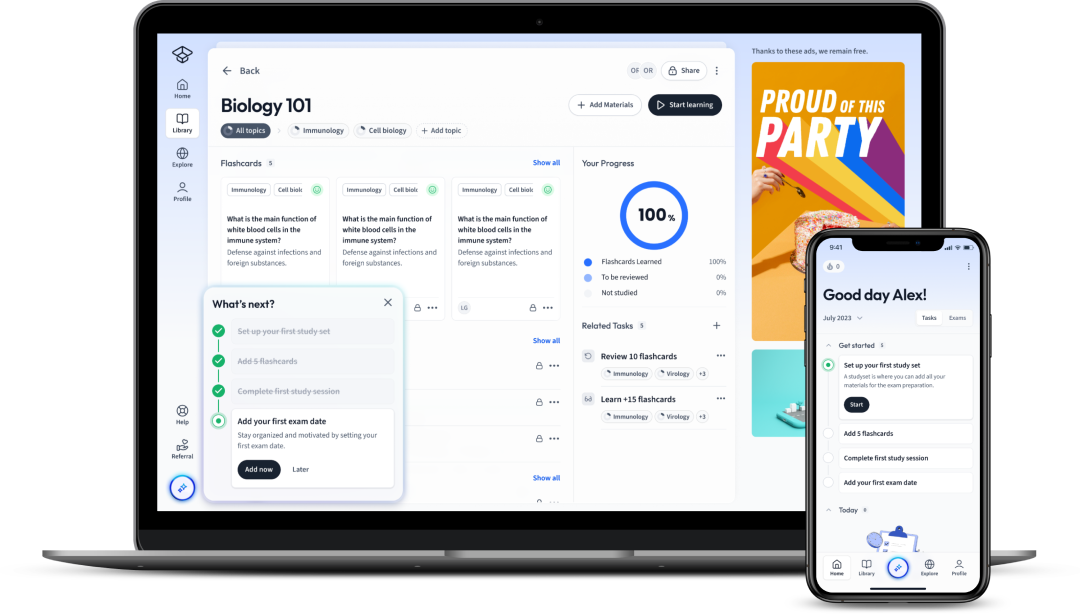
Explore our app and discover over 50 million learning materials for free.


























Lerne mit deinen Freunden und bleibe auf dem richtigen Kurs mit deinen persönlichen Lernstatistiken
Jetzt kostenlos anmeldenThere is a continuous cycling of materials in the ecosystem. In the environment, waste and dead materials are continually recycled to support various life forms. This is not just limited to organic materials. Both organic and inorganic minerals are continuously exchanged and moved in the Ecosystems to support the production of matter. This article will discuss the three most essential mineral cycles and their importance in ecosystems.
Organic and inorganic materials are continually recycled and reused. This cycle is commonly referred to as the nutrient cycle.
The nutrients in soil and water are taken up and used by Animals and Plants to grow and make new matter. After they die, their bodies decompose, releasing these nutrients back into the ecosystem.
Nutrient Cycles involve materials being converted to different forms that other organisms can use along the way. In Nutrient Cycles, elements are also moved to environments that are more accessible to living organisms. For instance, nitrogen is the most abundant gas in the atmosphere, but it is inert since living organisms cannot use it in the gaseous state. During the Nitrogen cycle, however, nitrogen is converted to more accessible forms by Bacteria that reside in the soil.
Microorganisms carry out the bulk of the processing of compounds in most nutrient cycles. They are integral to the biogeochemical cycle of nutrients in the soil by interconverting various compounds of nitrogen, carbon, and other elements. We will explain the roles of microorganisms in more detail as we discuss specific carbon, nitrogen and water cycles.
Biogeochemical cycles involve the circulation of nutrients between the living and non-living components of the ecosystem.
Without these cycles (and most importantly, decomposing microorganisms), essential nutrients would become trapped in the ground and the ocean, and life would cease to exist.
Carbon is the building block of all forms of life on earth. It is the central element for making complex biomolecules such as sugars, Proteins and DNA. Carbon is also found in the inorganic form of carbon dioxide (CO2) in the atmosphere. CO2 is a greenhouse gas essential for keeping the earth warm and maintaining its temperature suitable for life. Without CO2, the earth would be frozen solid. Too much CO2 in the atmosphere can also be problematic, as it can raise the average temperature beyond the optimum range and cause extreme weather events.
The carbon cycle describes the natural exchange of carbon atoms in different forms between the atmosphere, living organisms and the soil. Because our beloved planet and its atmosphere form a closed system, the amount of carbon in this system is always the same. It is the location of carbon atoms that continually changes. The carbon cycle has five main stages: Photosynthesis, decomposition, Respiration, combustion, and exchange.
Photosynthesis allows producers to uptake the carbon dioxide in the air and is essential in maintaining an oxygenated environment and cycling carbon. They use water, carbon dioxide and the energy from sunlight to produce high-energy Organic Molecules.
Photosynthesis is the process through which Plants convert sunlight, water, and carbon dioxide into oxygen and energy stored in the form of glucose.
Photosynthesis provides a source of energy for all living organisms. Photosynthetic producers, such as plants on land and phytoplankton in the ocean, reside at the bottom of the food chain in most ecosystems, which is why they are called primary producers.
Decomposition or Decay breaks down dead organic materials into simpler organic or inorganic molecules such as carbon dioxide, water, simple sugars, and Inorganic Ions.
Decay is an essential component of the nutrient cycle, responsible for recycling dead organic matter into nutrients that other living organisms, usually plants, can reuse. After an organism dies, its body starts to degrade quite rapidly. Decomposing microorganisms play a vital role at this stage, as they break down complex Biological Molecules into simpler products. This way, the carbon stored as Biomass is returned to the environment.
Saprotrophic Bacteria can be either aerobic (requiring oxygen) or anaerobic (working without oxygen). Aerobic decomposers release nutrients more efficiently than anaerobic decomposers, which release large amounts of carbon dioxide and methane as by-products.
Saprotrophs are simply decomposing microorganisms, such as bacteria and Fungi.
Over millions of years, dead inorganic matter (which has not been decomposed) will form fossil fuels under the pressure and heat from the earth's crust. These fossil fuels serve as a carbon sink, storing carbon away from the atmosphere.
Animals are unable to perform photosynthesis. So, they rely on obtaining energy from the food they ingest through cellular Respiration.
Plants also undergo respiration without light when they cannot perform photosynthesis.
Respiration is the opposite reaction to photosynthesis. It involves releasing the energy stored in glucose by using oxygen to convert it to carbon dioxide and water. The carbon dioxide released can then be used for photosynthesis.
Combustion is the process of burning something. Combustion and respiration are very similar to each other, and they both involve the use of oxygen to release energy and produce carbon dioxide and water. While the only substrate for respiration is glucose, all organic materials that contain carbon can undergo combustion.
By burning the wood that has been cut from a tree or petrol obtained from fossil fuels, the carbon stored in them is released into the atmosphere. Therefore, combustion plays a vital role in the carbon cycle, despite often being quite alarming!
Fossil fuels are formed very slowly over thousands of years. But humans have been burning fossil fuels much faster than the rate they are created. This has resulted in the release of carbon stored for thousands of years into the atmosphere and has significantly contributed to global warming.
Learn more by viewing our article on Global Warming here!
Another critical stage in the carbon cycle is exchange. Carbon dioxide and water continually circulate in the environment and interact with one another. CO2 from the atmosphere can dissolve in the oceans and form carbonic acid. This product of carbon dioxide and water is a weak acid, essential for maintaining the oceans' pH, making it suitable for marine organisms. If too much carbon dioxide is released into the environment, more will dissolve, making the oceans more acidic. Organisms that live in the oceans are susceptible to the pH of the water, and changes can significantly impact marine Biodiversity.
Ocean acidification can occur when the pH of seawater drops too low. Acidic conditions will put marine organisms under stress and reduce carbonate levels. Innumerable calcifying organisms require these carbonates to build up their calcareous shells and exoskeletons, so they struggle to survive in acidic conditions.
Nitrogen is an essential element for living organisms found in the environment in organic and inorganic forms.
Organic compounds of nitrogen are DNA, RNA, Proteins and amino acids.
Inorganic compounds of nitrogen are ammonia, nitrates and nitrites.
The Nitrogen cycle describes the exchange of nitrogen between various organic and inorganic forms as it is passed between the atmosphere and land-based ecosystems. The interconversion of nitrogen compounds occurs through both biological and physical processes. Four main steps are involved in the nitrogen cycle: nitrogen fixation, ammonification, nitrification, and denitrification.
Nitrogen gas (N2) is abundantly present in the atmosphere but is inaccessible to plants and animals while in the
Atmospheric nitrogen fixation occurs during lightning strikes. The high energy released during a lightning strike can break the bonds in nitrogen and oxygen gases and form nitrogen oxide. This new gas can then dissolve in the moisture in the air and convert to nitrates that fall with the rain.
Most nitrogen fixation is done by specific bacteria called diazotrophs in the soil. They contain a special enzyme called nitrogenase that catalyses the reaction between nitrogen gas and hydrogen atoms in water to form ammonia, which is subsequently converted to nitrates. Some diazotrophs symbiotically live in the root nodules of legumes. They fix nitrogen for the plant and, in exchange, receive Carbohydrates from it.
Because of this feature, legumes are used to increase the nitrogen content of nitrogen-poor soils.
Plants use the nitrates from nitrogen fixation to form complex Biological Molecules needed to grow. Herbivores then eat them. This way, the fixed nitrogen is passed on to animals.
The nitrogen in animals and plants is in the organic form of complex biological molecules. When these organisms release their waste products or after they die, the nitrogen present in them is still going to be organic. Bacteria and Fungi break down these complex organic compounds into inorganic ammonium in an ammonification process.
Plants can uptake ammonia but are unable to store it. Too much ammonia can be toxic for plants, so ammonia needs to be converted to nitrates to be usable. This process is called nitrification.
Nitrifying bacteria contain Enzymes that catalyse the oxidation of ammonia to nitrites. Another group of bacteria then further oxidise nitrites to nitrates. Nitrates can then be taken up by plants and used to build complex Organic Molecules.
Nitrates have a high solubility and form negatively charged ions when dissolved in water. Due to this, nitrates can enter the groundwater and reach rivers or drinking reservoirs. Elevated nitrate levels in drinking water are a concern since nitrates interfere with Blood oxygen levels in newborns.
While nitrification describes ammonia oxidation to nitrates, denitrification reduces nitrates to nitrites and eventually to nitrogen gas. By doing the opposite of nitrogen fixation, denitrification completes the nitrogen cycle.
Specific Types of Bacteria use the nitrate in the soil and convert it to nitrogen gas. These bacteria live deep in the soil and favour anaerobic conditions, such as waterlogged grounds.
Water is another material constantly being exchanged between different states and locations. The water molecule that falls with the rain today might have been flowing in a river yesterday or have been in the arctic ice cap thousands of years ago. This is due to water being able to change states at relatively small temperature differences.
Water is essential to all lifeforms on earth since it is the primary substrate of many reactions in living organisms. The water cycle ensures that fresh water is distributed globally so all living organisms can access safe, usable water.
The water cycle has four major stages: Evaporation, condensation, precipitation, and collection.
Evaporation describes the transition of water from a liquid state to a gas. The heat from the sun provides the energy for this transition. Water vapour is also released from the Transpiration of plants.
These vapours rise into the air and join one another to form clouds.
As the water vapour in the air rises, it cools down and condenses back into liquid form.
As water droplets accumulate and merge, they get bigger and heavier. They eventually begin to fall with the rain or snow. This is called precipitation.
The water that falls with rain or snow flows on the ground and joins with the rivers. Or it seeps into the ground and enters the underground water. Eventually, rivers and groundwater flow to lakes and oceans, where water is collected. The oceans freeze in cold environments such as the earth's poles to form ice caps. They are also collections of water that can be released after melting.
Water can then evaporate from these collection points to complete the cycle.
The nutrient cycle is a continuous process through which materials are recycled and reutilised. This cycle involves the passage of nutrients between Cells, organisms, Communities, and ecosystems.
Microorganisms carry out the bulk of the processing of compounds in most nutrient cycles.
Three primary cycles of materials are the carbon cycle, the nitrogen cycle, and the water cycle.
The carbon cycle consists of five main stages: photosynthesis, decomposition, respiration, combustion, and exchange.
The nitrogen cycle can be broken down into four main stages: nitrogen fixation, ammonification, nitrification, and denitrification.
As water cycles between different states and locations, it undergoes four main phases: evaporation, condensation, precipitation, and collection.
Nutrient cycles involve materials being converted to different forms that can be used by different organisms along the way. In nutrient cycles, elements are also moved to environments that are more accessible to living organisms. For instance, nitrogen gas is the most abundant gas in the atmosphere, but it is inert since living organisms cannot use it while it is in the gaseous state. During the nitrogen cycle, however, nitrogen is converted to more accessible forms by bacteria that reside in the soil.
The nutrients found in soil and water are taken up and used by animals and plants to grow and make new matter. After they die, their body decomposes and these nutrients are released back into the ecosystem.
Fossil fuels are formed very slowly over thousands of years. But humans have been burning fossil fuels much faster than the rate they are formed at. This has resulted in the release of carbon that had been stored for thousands of years into the atmosphere and has significantly contributed to global warming.
Three main cycles of materials are: carbon cycle, nitrogen cycle, and water cycle
Microorganisms carry out the bulk of the processing of compounds in most nutrient cycles. They are integral to the biogeochemical cycle of nutrients in the spoil by interconverting various compounds of nitrogen, carbon, and other elements.
The first learning app that truly has everything you need to ace your exams in one place
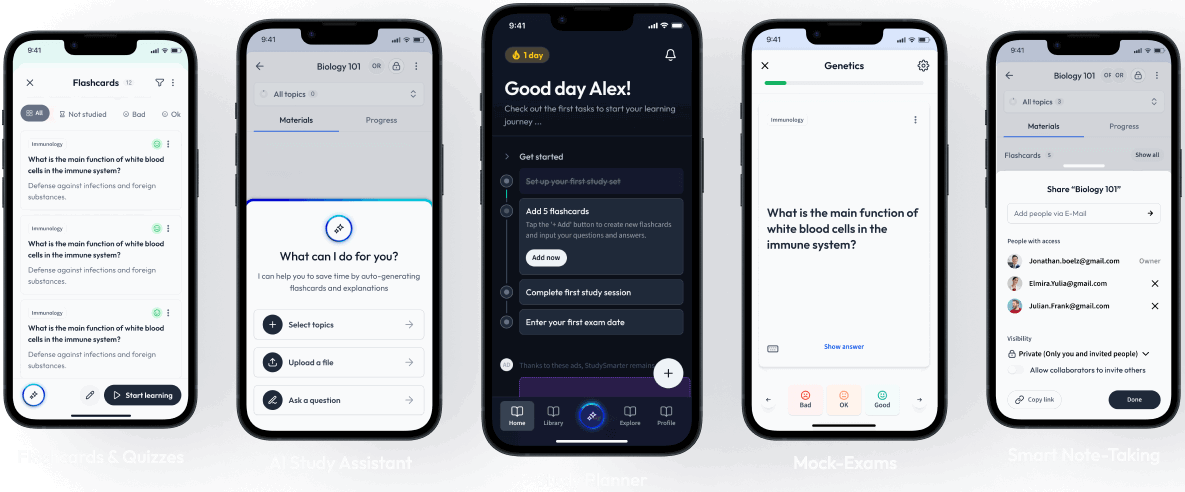
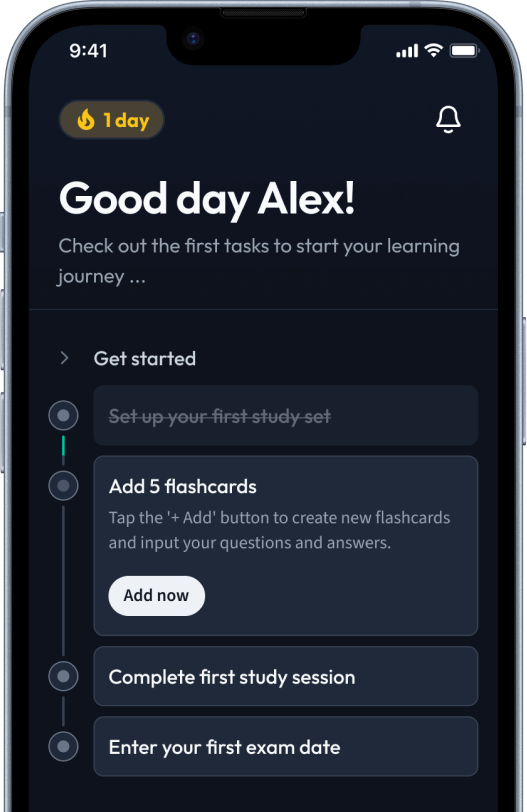
Sign up to highlight and take notes. It’s 100% free.
Save explanations to your personalised space and access them anytime, anywhere!
Sign up with Email Sign up with AppleBy signing up, you agree to the Terms and Conditions and the Privacy Policy of StudySmarter.
Already have an account? Log in