
StudySmarter - The all-in-one study app.
4.8 • +11k Ratings
More than 3 Million Downloads
Free
Americas
Europe
Carbohydrates, lipids, proteins, and nucleic acids are four biological macromolecules that are essential in sustaining life. Except for lipids, one thing these macromolecules have in common is that they are polymers made up of small identical monomers.
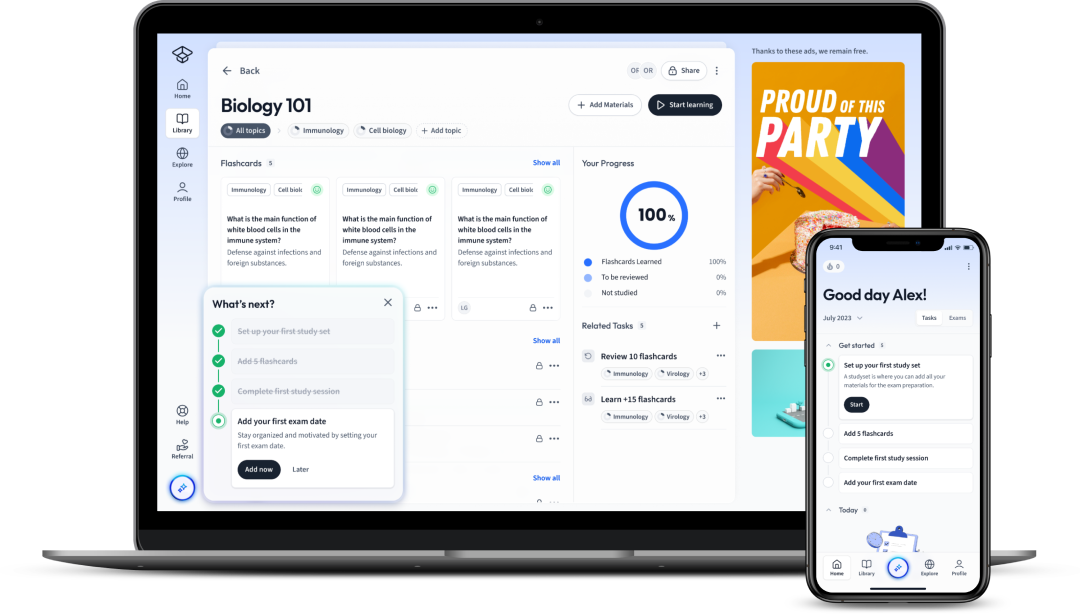
Explore our app and discover over 50 million learning materials for free.


























Lerne mit deinen Freunden und bleibe auf dem richtigen Kurs mit deinen persönlichen Lernstatistiken
Jetzt kostenlos anmeldenCarbohydrates, lipids, proteins, and nucleic acids are four biological macromolecules that are essential in sustaining life. Except for lipids, one thing these macromolecules have in common is that they are polymers made up of small identical monomers.
In the following we will define polymers, discuss the different types of polymers, and cite various examples of each type. We will also discuss several examples of artificial or synthetic polymers and how they are typically used.
Let's start by looking at the definition of a polymer.
Polymers are large, complex molecules that are made up of simpler, smaller identical subunits called monomers.
It is helpful to remember that the prefix “poly-” means “many”. A polymer is made up of many monomers! It is also helpful to consider a polymer to be a chain of repeating monomer units.
Think train: each car is a monomer, and the whole train, which consists of identical cars, is the polymer.
To form a polymer, monomers undergo a process called dehydration synthesis (which is also sometimes called condensation reaction).
Dehydration synthesis is where monomers are joined together by covalent bonds and a water molecule is released as a by-product (Fig. 1).
Polymer molecules are joined by covalent bonds that are specific to each type of polymer which we will discuss in further detail later.
On the other hand, the covalent bonds that link polymers can be broken down by adding water through a process called hydrolysis (Fig. 2). Hydrolysis is basically the opposite of dehydration synthesis.
During hydrolysis, the covalent bonds that link polymers can be broken down by the addition of water.
The hydrolysis of each polymer is catalyzed by a specific enzyme. We will also discuss this in further detail later as we go through each type of polymer.
'Dehydration' literally means the removal or loss of water, while 'synthesis' means the combination of molecules or substances.
A covalent bond is a type of chemical bond formed between atoms that share valence electrons.
The majority of biological macromolecules are made up of six elements in various amounts and configurations:
Here, we will discuss the types of polymer biological macromolecules (carbohydrates, proteins, and nucleic acids) and their monomer precursors. We will also discuss how they are formed and broken down. We will also discuss why lipids are not considered polymers.
Carbohydrates are chemicals that give living organisms energy and structural support. Based on the amount of monomers in the macromolecule, carbohydrates are categorized into monosaccharides, disaccharides, and polysaccharides.
Monosaccharides make up carbohydrate molecules. Each monosaccharide molecule contains only three elements:
Examples of monosaccharides include glucose, galactose, and fructose. When monosaccharides combine, they form carbohydrate polymers which are held together by a type of covalent bond called glycosidic bonds. Carbohydrate polymers include disaccharides and polysaccharides.
Disaccharides are polymers composed of two monosaccharides. Examples of disaccharides include maltose and sucrose. Maltose is produced through the combination of two monosaccharides molecules. It is more commonly referred to as malt sugar. Sucrose is produced through the combination of glucose and fructose. Sucrose is also known as table sugar.
Polysaccharides are polymers composed of three or more monosaccharides. Complex carbohydrates are polysaccharides: starch, glycogen, and cellulose. All three are composed of repeating units of glucose monomers.
Carbohydrates are broken down by enzymes that are specific to the molecule. For example, maltose is broken down by the enzyme maltase, while sucrose is broken down by the enzyme sucrase.
Proteins are biological macromolecules that serve a variety of roles, including structural support and serving as enzymes to catalyze biological events. Examples of proteins include hemoglobin and insulin. Proteins consist of amino acid monomers.
Each amino acid molecule has:
A carbon atom
An amino group (NH2)
A carboxyl group (COOH)
A hydrogen atom
Another atom or organic group referred to as the R group
There are 20 commonly used amino acids, each with its own R group. Amino acids differ in their chemistry (acidity, polarity, and so on) and structure (helices, zigzags, and other shapes).
When amino acids undergo dehydration synthesis, they form polypeptides that are held together by peptide bonds. A protein molecule has at least one polypeptide chain. Protein function and structure differ depending on the type and sequence of the amino acid monomers.
The peptide bonds in proteins are hydrolyzed by the enzymes peptidase and pepsin with the help of hydrochloric acid.
Nucleic acids are complex molecules that store genetic information and instructions for cellular functions. The two most essential nucleic acids are ribonucleic acid (RNA) and deoxyribonucleic acid (DNA).
Nucleic acids are polymers that consist of nucleotide monomers. Each nucleotide has three major components:
A nitrogenous base
A pentose (five-carbon) sugar
A phosphate group
A phosphodiester bond connects one nucleotide to another nucleotide. It is formed when the phosphate group links the pentose sugars of adjacent nucleotides. Because the pentose sugar and the phosphate group produce a repetitive, alternating pattern, the resulting structure is called the sugar-phosphate backbone.
RNA is a single stranded nucleic acid molecule, while DNA is a double stranded molecule where the two strands are held together by hydrogen bonds.
DNA can be hydrolyzed by enzymes called nucleases. On the other hand, RNA can be hydrolyzed by enzymes called ribonucleases.
A hydrogen bond is a type of intramolecular attraction between one molecule's partially positive hydrogen atom and another molecule's partially negative atom.
Fats, steroids, and phospholipids are among the nonpolar biological macromolecules known as lipids. Lipids consist of a combination of fatty acids and glycerol.
Fatty acids are long hydrocarbon chains with a carboxyl group (COOH) at one end. A hydrocarbon chain is an organic molecule made up of carbon and hydrogen atoms linked together in a chain.
When fatty acids combine with glycerol, they form glycerides:
One fatty acid molecule attached to a glycerol molecule forms a monoglyceride.
Two fatty acid molecules attached to a glycerol molecule form a diglyceride.
While these glycerides are prefixed with mono- and di- just like the saccharides, they are not considered polymers. This is because the fatty acids and glycerol units contained in lipids vary in amount, meaning they form a chain with dissimilar, non-repeating units.
A nonpolar molecule is one whose atoms have equal electronegativity and thus share electrons equally.
We have discussed the polymer molecules that are essential to life. But not all polymers are naturally occurring in nature: some of them are artificially created by humans. Such artificial or synthetic polymers include polyethylene, polystyrene, and polytetrafluoroethylene.
While these names make them sound like things you can only find in science labs, these are actually materials you would encounter in your everyday life.
Polyethylene is a transparent, crystalline, and flexible polymer. Its monomer is ethylene (CH2=CH2).
Polyethylene has two widely-used forms: low-density polyethylene (LDPE) and high-density polyethylene (HDPE). LDPE tends to be a soft and waxy solid material. It is used in manufacturing film wraps and plastic bags. On the other hand, HDPE tends to be a more rigid material. It is typically used in electrical insulation, plastic bottles, and toys.
While they are made of the same monomers, the masses of HDPE and LDPE are vastly different: synthetic HDPE macromolecules range from 105 to 106 amu (atomic mass unit) whereas LDPE molecules are more than a hundred times smaller.
Polystyrene is a hard, rigid, clear solid material that can be dissolved in organic solvents. It is a synthetic polymer made up of styrene monomers (CH2=CHC6H5). It is popularly used in the food industry in the form of disposable plates, trays, and beverage cups.
Polytetrafluoroethylene is a synthetic polymer that is made of tetrafluoroethylene monomers (CF2=CF2). This material exhibits excellent resistance to heat and chemicals, which is why it is commonly used in electrical insulation. It is also the material used to give cooking ware a non-stick surface.
Polymers are large, complex molecules that are made up of simpler, smaller identical subunits called monomers.
Carbohydrates, proteins, and nucleic acids are some naturally-occurring polymers essential to life. Polyethylene and polystyrene are examples of synthetic polymers used in our everyday lives.
Yes, DNA is a polymer consisting of nucleotide monomers.
There are 4 types of biological macromolecules that are essential to life: carbohydrates, proteins, lipids, and fatty acids. With the exception of lipids, these are all polymers.
Lipids are not considered polymers because they are made of dissimilar and non-repeating units consisting of fatty acids and glycerol in varying quantities.
Flashcards in Polymer43
Start learningExplain how monomers form polymers.
To form a polymer, monomers undergo a process called dehydration synthesis (which is also sometimes called condensation reaction). Dehydration synthesis is where monomers are joined together by covalent bonds and a water molecule is released as a by-product. Such covalent bonds are specific to each type of polymer.
Explain how polymers are broken down into monomers.
The covalent bonds that link polymers can be broken down by adding water through a process called hydrolysis. The hydrolysis of each polymer is catalyzed by a specific enzyme.
Which among the four essential biological macromolecules is not considered a polymer?
Lipids
When monosaccharides combine, they form carbohydrate polymers which are held together by a type of covalent bond called ______.
Glycosidic bonds
Sucrose is a disaccharide consisting of what monosaccharides?
Glucose
What is the relationship between a protein molecule and a polypeptide?
A polypeptide is a chain of amino acids held together by peptide bonds. A protein molecule consists of one or more polypeptide chains.
Already have an account? Log in
Open in AppThe first learning app that truly has everything you need to ace your exams in one place
Sign up to highlight and take notes. It’s 100% free.
Save explanations to your personalised space and access them anytime, anywhere!
Sign up with Email Sign up with AppleBy signing up, you agree to the Terms and Conditions and the Privacy Policy of StudySmarter.
Already have an account? Log in